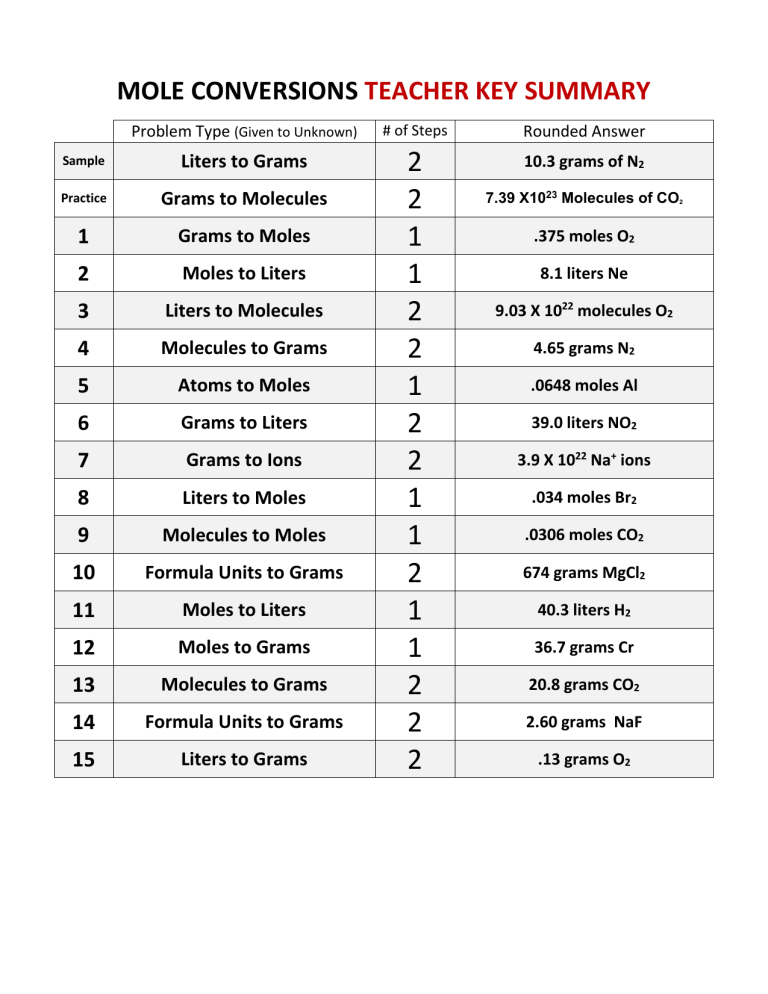
Molarity Molarity = grams / molar mass / Liters Liters = grams / molar mass/ Molarity Grams = Molarity x Liters x molar mass. - ppt download

metric meterlitergramwkst copy.pdf - Name Date Per Writing Meters Liters and Grams When Moving from one unit to a larger unit No. of units moved Unit | Course Hero

SOLVED:Write a unit conversion factor to convert a. meters to kilometers b. grams to centigrams c. liters to milliliters
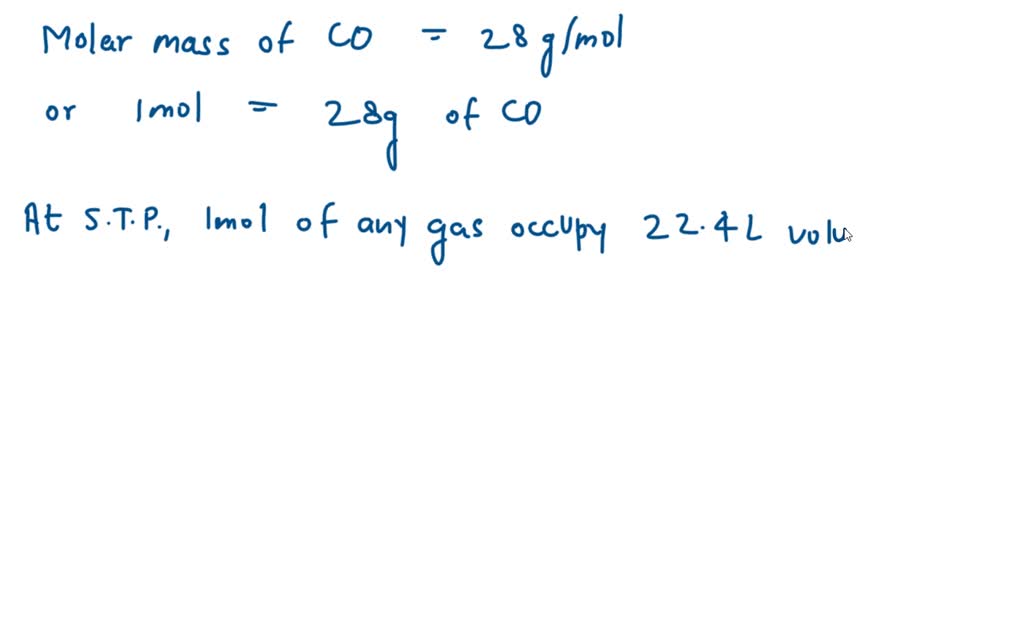
SOLVED: What is the mass in grams of 1 liter of carbon monoxide, CO, at standard temperature and pressure, STP? Note the molecular weight of CO is 28 g/mole and at STP,
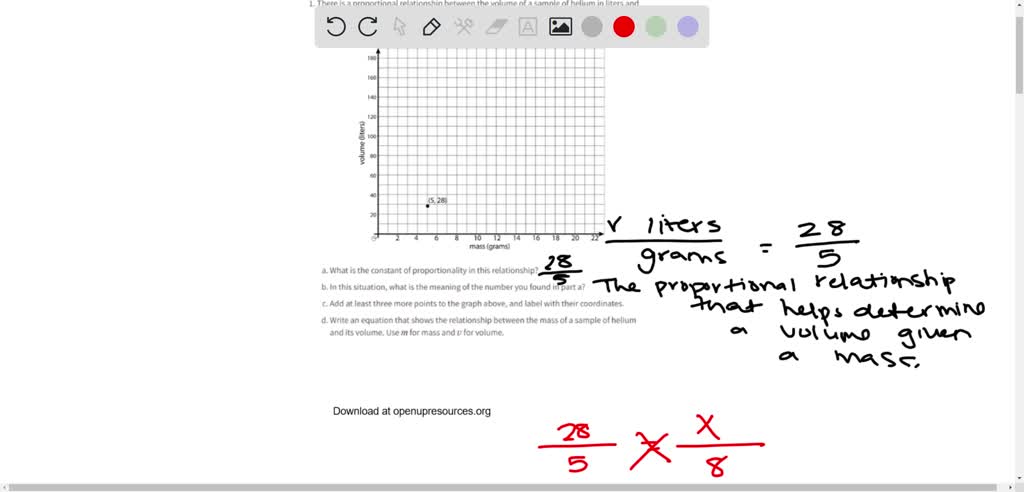
SOLVED:There is a proportional relationship between the volume of a sample of helium in liters and the mass of that sample in grams. If the mass of a sample is 5 grams,














![Learn Metric Units & Unit Conversions (Meters, Liters, Grams, & more) - [5-8-1] - YouTube Learn Metric Units & Unit Conversions (Meters, Liters, Grams, & more) - [5-8-1] - YouTube](https://i.ytimg.com/vi/T8WZU1QqSgI/maxresdefault.jpg)